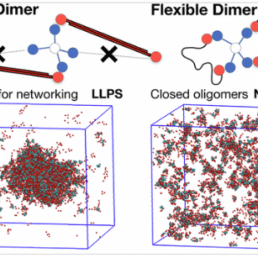
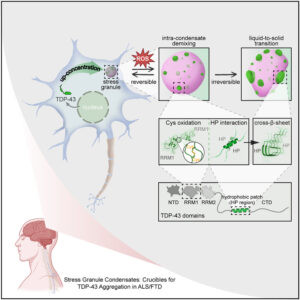
Intra-condensate demixing of TDP-43 inside stress granules generates pathological aggregates
A new study from the labs of Honigmann, Hyman, and Alberti in Dresden, in addition to colleagues in Texas A&M University, Mayo Clinic, Brown University, and Saint Louis University investigates the mechanism behind pathological outcomes of protein aggregation inside stress granules. The authors of the study entitled “Intra-condensate demixing of TDP-43 inside stress granules generates pathological aggregates” and published in Cell in May, 2025, determined that aggregation of TAR DNA-binding protein 43 (TDP-43) is induced by two events, namely up-concentration of TDP-43 in stress granules beyond a threshold and oxidative stress and described the mechanism behind the observation. They use this new understanding to engineer TDP-43 variants resistant to aggregation in the cell.
Impact: The aggregation of TDP-43 in motor neurons is a hallmark of neurodegenerative diseases including amyotrophic lateral sclerosis (ALS). Understanding the mechanisms leading to aggregation paves the path towards developing preventive and therapeutic strategies.
Citation:
Yan, X., Kuster, D., Mohanty, P., Nijssen, J., Pombo-García, K., Garcia Morato, J., Rizuan, A., Franzmann, T. M., Sergeeva, A., Ly, A. M., Liu, F., Passos, P. M., George, L., Wang, S.-H., Shenoy, J., Danielson, H. L., Ozguney, B., Honigmann, A., Ayala, Y. M., Fawzi, N. L., Dickson, D. W., Rossoll, W., Mittal, J., Alberti, S., & Hyman, A. A. (2025). Intra-condensate demixing of TDP-43 inside stress granules generates pathological aggregates. Cell, 188(15), 4123-4140.e4118. https://doi.org/10.1016/j.cell.2025.04.039