Phase separation in cell polarity
In this video abstract, Shamba Saha tells you about his recent publication in Cell, “Polar Positioning of Phase-Separated Liquid Compartments in Cells Regulated by an mRNA Competition Mechanism.” Learn more in the full paper Saha et al. (2016) published in Cell, http://dx.doi.org/10.1016/j.cell.2016…
Written and narrated by Shamba Saha. Edited and produced by Lisa Dennison.
Regulation of centrosome assembly by phosphorylation
In this video abstract, Oliver Wueseke tells you about his recent publication in Biology Open, “Polo-like kinase phosphorylation determines C.elegans centrosome size and density by biasing SPD-5 toward an assembly-competent conformation,”
Learn more in the full paper Wueseke et al. (2016) http://dx.doi.org/10.1242/bio.020990
Written and narrated by Oliver Wueseke, illustrated by Jeff Woodruff, edited and produced by Lisa Dennison.
Building a synthetic centrosome
The centrosome is a structure in cells which is built every time the cell prepares to divide. In this video abstract, Jeff Woodruff explains how to build a centrosome in a test tube from purified proteins. He found that the centrosome acts like a sponge, which selectively soaks up the proteins needed to build microtubules.
Read more in the full paper, published in the journal Cell: http://www.cell.com/cell/fulltext/S00…
See the full blog post at: http://www.sciencesketches.org/single…
Jeff Woodruff is a postdoc in the lab of Tony Hyman (hymanlab.org) at the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden, Germany
Microtubule nucleation through phase separation
Cells must build cytoskeleton structures, such as microtubules, where they are locally needed within the cell, but how do they do this? In this video abstract, Amayra Hernández-Vega explains that in a test tube, microtubules can be formed locally through phase separation. She also found that the protein Tau can phase separate into droplets, which can then concentrate enough tubulin to nucleate and build microtubules. Read more in the full paper, published in the journal Cell Reports: http://www.cell.com/cell-reports/abst… and see the full blog post at: http://www.sciencesketches.org/single…
Amayra Hernández-Vega is a postdoc in the lab of Tony Hyman (http://hymanlab.org) at the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden, Germany.
A role for RNA in Stress Granules assembly
Stress granules are membraneless compartments formed by phase separation of specific molecules upon exposure to cellular stress such as oxidative stress, heat shock, or osmotic stress.
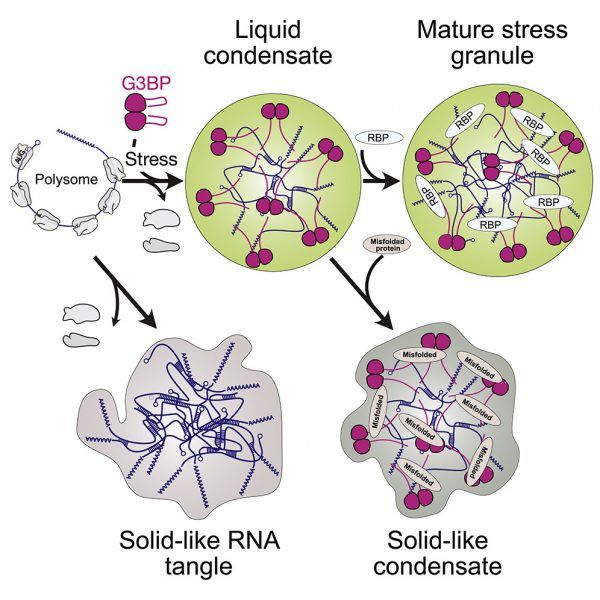
The Alberti, Jahnel, Honigmann, and Hyman labs published a study in cell highlighting the role of RNA in the assembly of stress granules by crosslinkinig with G3BP clusters and how G3BP clusters in return prevent RNA entanglement. The study entitled "RNA-Induced Conformational Switching and Clustering of G3BP Drive Stress Granule Assembly by Condensation" is in collaboration with Washington University, the European Molecular Biology Laboratory, Heidelberg, and Pohang University of Science and Technology, Korea.
Graphical Abstract:
Stressed cells shut down translation, release mRNA molecules from polysomes, and form stress granules (SGs) via a network of interactions that involve G3BP. Here we focus on the mechanistic underpinnings of SG assembly. We show that, under non-stress conditions, G3BP adopts a compact auto-inhibited state stabilized by electrostatic intramolecular interactions between the intrinsically disordered acidic tracts and the positively charged arginine-rich region. Upon release from polysomes, unfolded mRNAs outcompete G3BP auto-inhibitory interactions, engendering a conformational transition that facilitates clustering of G3BP through protein-RNA interactions. Subsequent physical crosslinking of G3BP clusters drives RNA molecules into networked RNA/protein condensates. We show that G3BP condensates impede RNA entanglement and recruit additional client proteins that promote SG maturation or induce a liquid-to-solid transition that may underlie disease. We propose that condensation coupled to conformational rearrangements and heterotypic multivalent interactions may be a general principle underlying RNP granule assembly.
Read the full publication
https://www.sciencedirect.com/science/article/pii/S0092867420303421
Filament formation by the translation factor eIF2B regulates protein synthesis in starved cells
Aminoacyl-tRNA synthetases (aaRSs), the enzymes responsible for coupling tRNAs to their cognate amino acids, minimize translational errors by intrinsic hydrolytic editing. Here, we compared norvaline (Nva), a linear amino acid not coded for protein synthesis, to the proteinogenic, branched valine (Val) in their propensity to mistranslate isoleucine (Ile) in proteins. We show that in the synthetic site of isoleucyl-tRNA synthetase (IleRS), Nva and Val are activated and transferred to tRNA at similar rates. The efficiency of the synthetic site in pre-transfer editing of Nva and Val also appears to be similar. Post-transfer editing was, however, more rapid with Nva and consequently IleRS misaminoacylates Nva-tRNAIle at slower rate than Val-tRNAIle. Accordingly, an Escherichia coli strain lacking IleRS post-transfer editing misincorporated Nva and Val in the proteome to a similar extent and at the same Ile positions. However, Nva mistranslation inflicted higher toxicity than Val, in agreement with IleRS editing being optimized for hydrolysis of Nva-tRNAIle. Furthermore, we found that the evolutionary-related IleRS, leucyl- and valyl-tRNA synthetases (I/L/VRSs), all efficiently hydrolyze Nva-tRNAs even when editing of Nva seems redundant. We thus hypothesize that editing of Nva-tRNAs had already existed in the last common ancestor of I/L/VRSs, and that the editing domain of I/L/VRSs had primarily evolved to prevent infiltration of Nva into modern proteins.
Keywords: aminoacyl-tRNA synthetase; mistranslation; non-proteinogenic amino acids; primordial translation; proofreading.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Condensation regulates translation
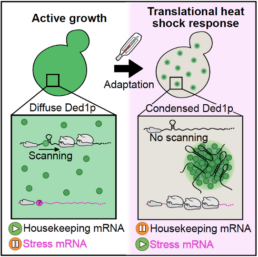
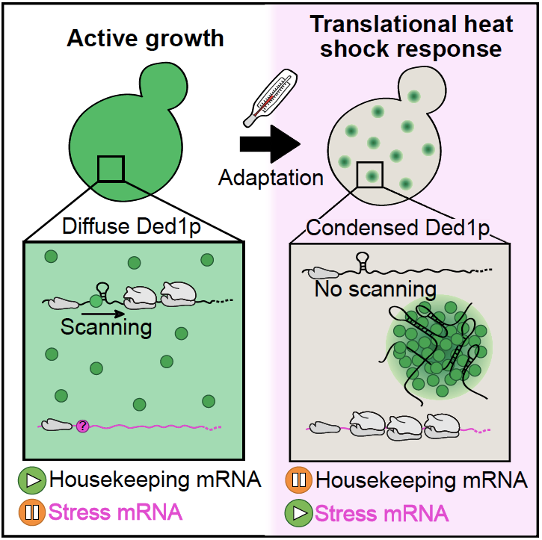
New insights into the influence of Ded1p condensation on translation comes from the Hyman, Alberti and Kreysing labs. The study published in Cell is entitled "Condensation of Ded1p Promotes a Translational Switch from Housekeeping to Stress Protein Production".
Graphical abstract:
Cells sense elevated temperatures and mount an adaptive heat shock response that involves changes in gene expression, but the underlying mechanisms, particularly on the level of translation, remain unknown. Here we report that, in budding yeast, the essential translation initiation factor Ded1p undergoes heat-induced phase separation into gel-like condensates. Using ribosome profiling and an in vitro translation assay, we reveal that condensate formation inactivates Ded1p and represses translation of housekeeping mRNAs while promoting translation of stress mRNAs. Testing a variant of Ded1p with altered phase behavior as well as Ded1p homologs from diverse species, we demonstrate that Ded1p condensation is adaptive and fine-tuned to the maximum growth temperature of the respective organism. We conclude that Ded1p condensation is an integral part of an extended heat shock response that selectively represses translation of housekeeping mRNAs to promote survival under conditions of severe heat stress.
The ALS-Associated FUS (P525L) Variant Does Not Directly Interfere with Microtubule-Dependent Kinesin-1 Motility
The Diez lab, in collaboration with the Hermann lab in Rostock, Germany, have utilised an in vitro microtubule gliding motility assay to study deficient intracellular transport in motor neurons. The findings exclude a role of mutations in the phase-separating FUS protein, known to be mutated in neurodegenerative diseases like amyotrophic lateral sclerosis (ALS). in The study was published in the International Journal of Molecular Sciences as part of the Special Issue Molecular Mechanisms of Aging-Related Neurodegenerative Diseases.

Cytoskeletal organization through multivalent interactions
Stefan Diez and collaborators Zdenek Lansky and Marcus Braun from the Institute of Biotechnology of the Czech Academy of Sciences, Czech Republic, published an Opinion article emphasizing the role of multivalent interactions cytoskeletal phenomena including (1) the generation of entropic forces by filament crosslinkers, (2) processivity of molecular motors, (3) spatial sorting of proteins, and (4) concentration-dependent unbinding of filament-associated proteins. The article was published in the Journal of Cell Science.
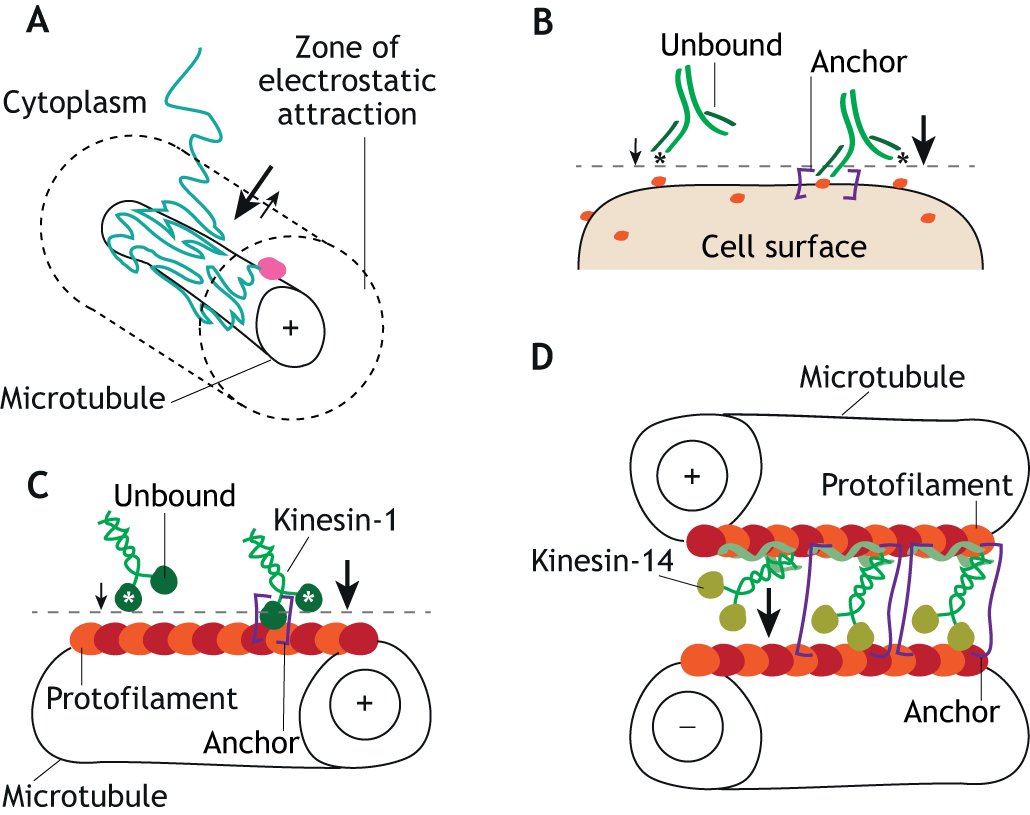 Consequences of multivalent interactions on ligand–receptor interaction kinetics
Consequences of multivalent interactions on ligand–receptor interaction kinetics
ELBE Postdoctoral Fellows Program
To apply for a postdoc position through the ELBE Postdoctoral Fellows Program in Dresden, visit the page of the Center for Systems Biology Dresden.