MPI-PKS Visitors Program
Guest scientist positions are available for nearly all academic career levels, ranging from PhD students, over young postdoctoral researchers, to senior scientists spending their sabbaticals at MPI-PKS. For more information, visit the MPI-PKS Visitors Program web page.
Kinetically distinct phases of tau on microtubules regulate kinesin motors and severing enzymes
The Diez and Hyman labs, in collaboration with the Lansky and Braun lab at the Institute of Biotechnology of the Czech Academy of Sciences, Czech Republic, published a study on the role of phase separation of the intrinsically disordered Tau protein in regulating the activity of motor proteins and severing enzymes on microtubules. The study, published in Nature Cell Biology demonstrated using in vitro protein reconstitution that Tau protects microtubules from degradation by forming reversible cohesive islands.

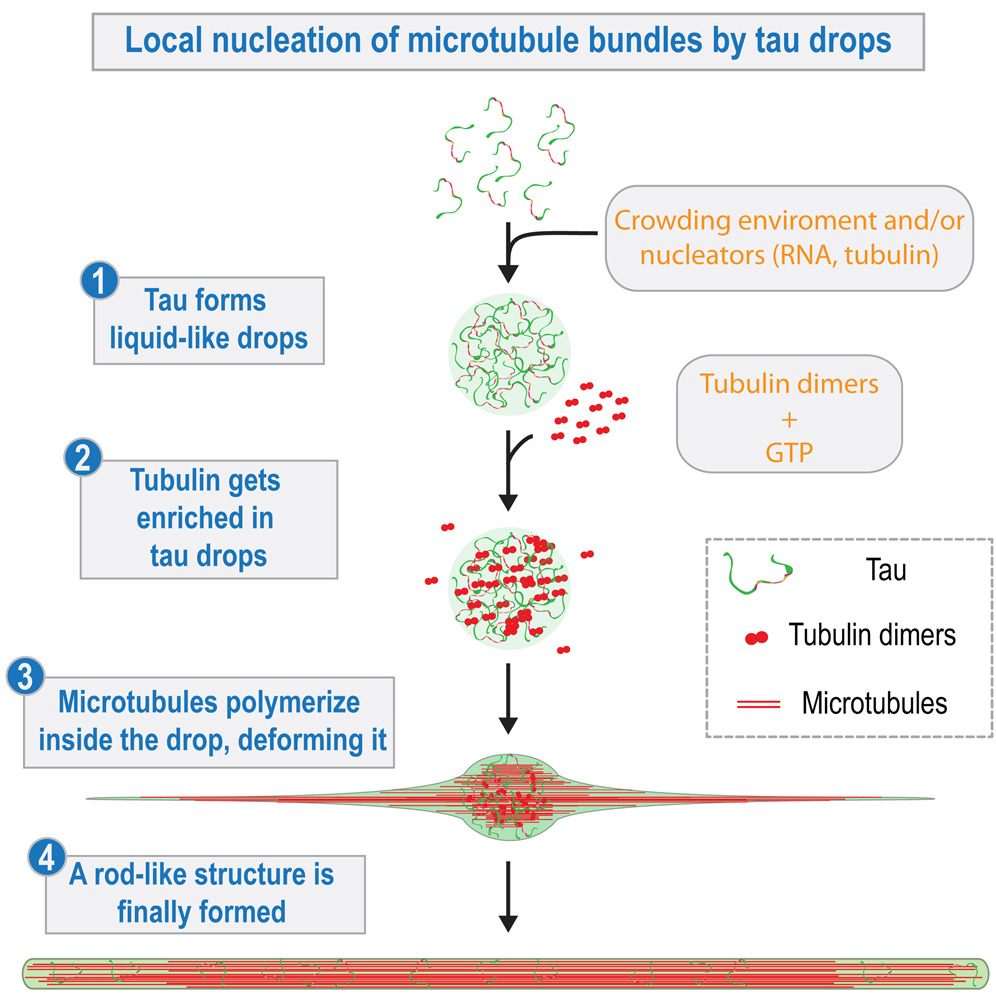
Local nucleation of microtubule bundles through tubulin concentration into a condensed tau phase
Theskeleton that supports the structure of our cells, termed cytoskeleton is formed of several kinds of polymers including actin and microtubules. How the single units on the polymers (monomers and dimers) are concentrated to gether to synthesize the polymer is the subject of this study by the labs of Diez, Hyman, and Alberti. The authors identified a protein associated to the cytoskeleton, named Tau, that is able to phase-separate from the cytosol. Tau concentrates the tubulin dimers, which in turn polymerize to make microtubules inside the tau droplet. As opposed to this physiological phenomenon, pathological aggregation of mutated tau is a hallmark of neurodegenerative disorders, including Alzheimer’s disease.
Read more: https://doi.org/10.1016/j.celrep.2017.08.042